News
Filter results

Announcements
Center for Development and Production of Highly Potent Drugs
TZF Polfa, a Polish pharmaceutical manufacturer, proudly presents its latest venture in Warsaw – the Center for Development and Production of Highly Potent Drugs, one of the most…
Read more
Uncategorized
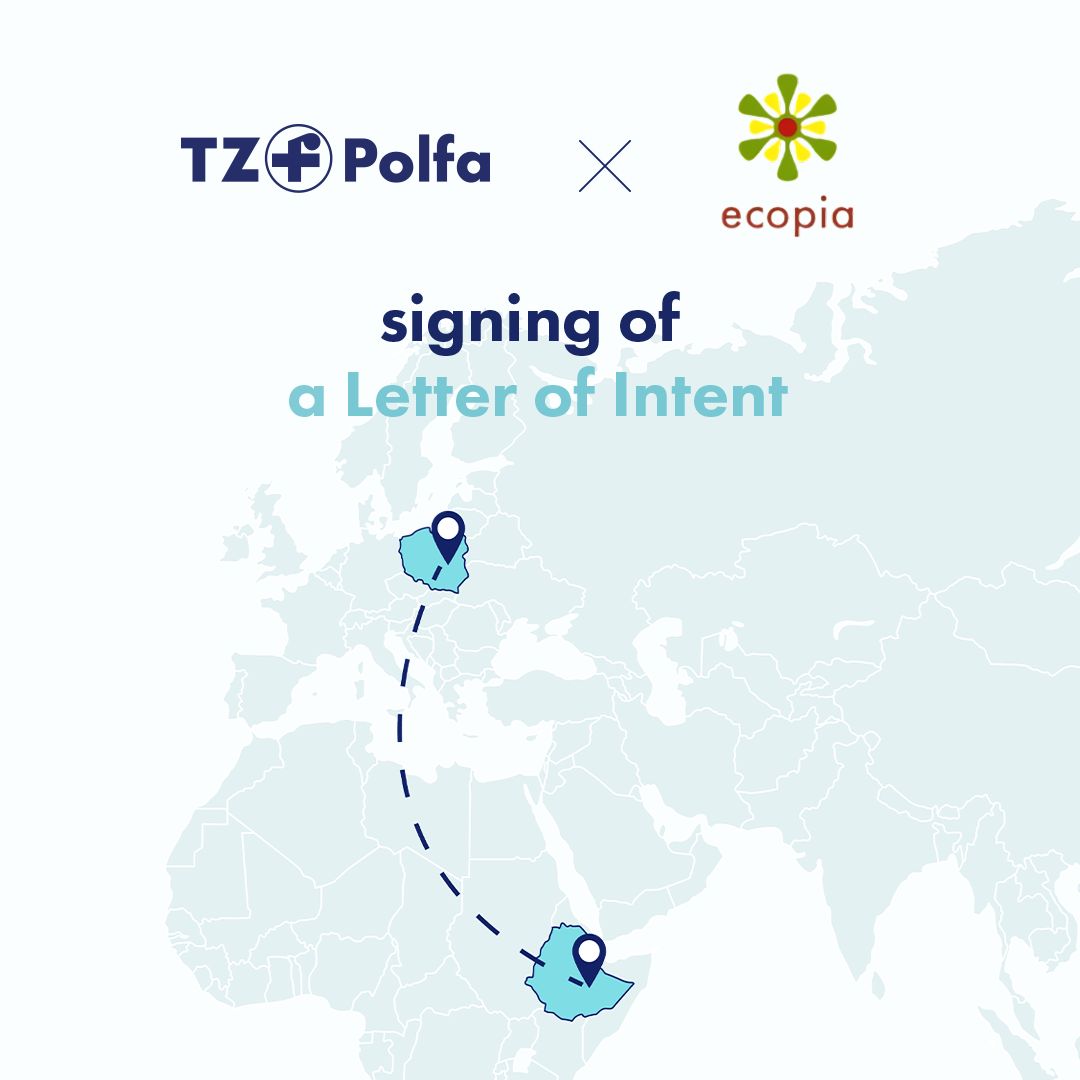
TZF Polfa and Ecopia – Cooperation to Enhance Access to Medicines in Ethiopia
TZF Polfa S.A. has signed a Letter of Intent (LOI) with Ecopia, initiating cooperation aimed at developing the pharmaceutical market in Ethiopia and improving access to critically important…
Read more

Tenders
Request fot quotatnion – RFQ Z2/5438/1 – Study monitoring – Empagliflozin
In connection with the implementation of the project 2022/ABM/04/00014-00 – „Opracowanie i rozwój produktu leczniczego z substancją czynną EMPAGLIFLOZYNĄ – leku generycznego na cukrzycę typu 2” (”Design and…
Read more

Announcements
Selection procedure for the position of President of the Management Board
The Supervisory Board of Tarchomińskie Zakłady Farmaceutyczne “Polfa” S.A., with its registered office in Warsaw, hereby announces a selection procedure for the position of President of the Management…
Read more

Announcements
Selection procedure for the position of First Member of the Management Board
The Supervisory Board of Tarchomińskie Zakłady Farmaceutyczne “Polfa” S.A., with its registered office in Warsaw, hereby announces a selection procedure for the position of First Member of the…
Read more

Announcements
Selection procedure for the position of Second Member of the Management Board
The Supervisory Board of Tarchomińskie Zakłady Farmaceutyczne “Polfa” S.A., with its registered office in Warsaw, hereby announces a selection procedure for the position of Second Member of the…
Read more

For patients
Availability of Elenium
Warsaw, January 20, 2026 Elenium Availability Dear Sir or Madam, Following the administrative decision of the Chief Pharmaceutical Inspectorate to withdraw from the market certain…
Read more
For patients
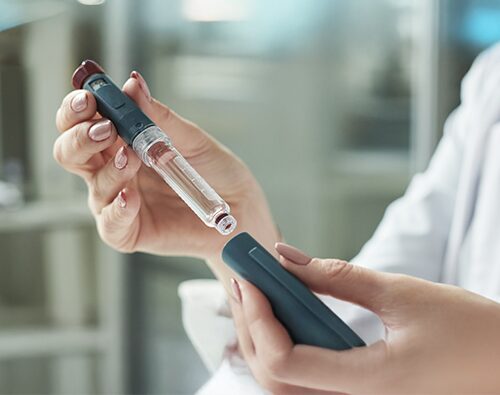
Distribution of PolhuminPen injectors
Dear Patients and Doctors, Please be advised that PolhuminPen® injectors are distributed free of charge exclusively as part of a patient program for individuals requiring insulin therapy. To…
Read more

Announcements
Success in the development of the medicinal product Dapagliflozinum 10 mg
We are pleased to announce that the newly developed medicinal product Dapagliflozinum, 10 mg, coated tablets, has successfully passed bioequivalence testing. The project is co-financed from the state…
Read more

